Worked out examples
Q.
The chief constituent in eucalyptus oil is a substance called eucalyptol, which contains 77.87% of C, 11.76% of Hydrogen and 10.37% of Oxygen
(a) What is the empirical formula for this substance?
(b)The mass spectrum of eucalyptol shows a peak at about 154 amu. What is the
molecular
formula ?
Sol.
(a) We now have the different compositions of ‘C’, ‘H’ and ‘O’ present in the sample.
In a chemical formula, the subscripts represent the ratio of
number of moles of each element that combine to form one mole of the compound.
If we
assume exactly 100g sample of the compound we know the mass of each element
in the compound.
Assume that we have 100g of eucalyptol containing 77.87 grams of
carbon, 11.76 grams of Hydrogen and 10.37grams of oxygen. As subscripts represent the mole
ratio, we need to convert the grams of each element to moles. The conversion factor needed
is the molar mass of each element.
Let n represent the number of moles of each element
so that
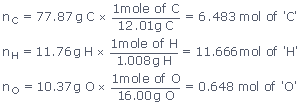
Now we arrive at a formula C6.483H11.66O0.648.
But as the chemical formulas are written in whole numbers we have to make this real
numbers to whole numbers.For this convert the real numbers to whole number divide
all the numbers by smallest subscript(0.648)
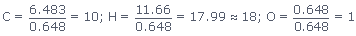
Now the emperical formula for
eucalyptol is C10H18O
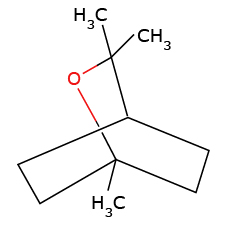
(b) As the mass spectrum of eucalyptol shows a peak at
about 154
amu.
Now the weight of
| C10H18O | = | 10 × 12.01 + 18 × 1.008 + 1 × 16.00 |
| = | 120.1 + 18.144 + 16 | |
| = | 154.244 |
Hence molecular formula wt = 154.244
Molecular formula is C10H18O