 John Newlands
In 1863, John Newlands, an English chemist, in his written paper has classified 56 elements into 11 groups
based on physical properties and proposed the Law of Octaves which states that every eighth element repeated its physical and
chemical properties.
John Newlands
In 1863, John Newlands, an English chemist, in his written paper has classified 56 elements into 11 groups
based on physical properties and proposed the Law of Octaves which states that every eighth element repeated its physical and
chemical properties.The musical analogy was ridiculed at that time, but was found to be insightful after the work of Mendeleev and Meyer were published.
Newlands law of Octaves :
After Dobereiner's ideas were ruled out, an English chemist John Alexander Reina Newlands in 1863–64 noted that every
eighth element showed similar physical and chemical properties, when the elements are placed in the increasing order of their
atomic masses. This was called the Newlands law of octaves. The law states that when elements are placed in the increasing order
of atomic masses, the properties of the eighth elements are repeated.
Newlands arranged the elements in the following manner:
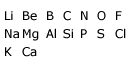
Row of elements had seven elements and the eighth fell under the first element. In those days, the number of elements known was very limited and no elements from the noble or inert gas elements such as helium (He), neon (Ne), argon (Ar), etc., were known.
 Newlands had arranged the known elements during his time according to the weight. His law is obeyed up to
calcium. But this law is not obeyed by elements with atomic weight greater than ‘Ca’. Newlands did not left any space
for the noble gas which was not known at that time.
Newlands had arranged the known elements during his time according to the weight. His law is obeyed up to
calcium. But this law is not obeyed by elements with atomic weight greater than ‘Ca’. Newlands did not left any space
for the noble gas which was not known at that time.
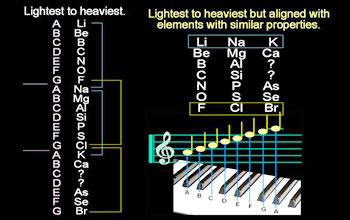
Let us observe elements in the first column. Li is the first element. The eighth element after Li is Na. Similarly, the eighth element after Na is K. So from the Newlands law of octaves, elements Li, Na and K will have similar chemical properties. All obey this law. All the elements are metallic, highly reactive and show a valency of +1. They are known as alkali elements.
On moving right, if we take beryllium (Be) as the first element, the eighth element from Be is magnesium (Mg). If we continue in the similar fashion, the eighth element after Mg is calcium (Ca). According to Newlands law, the elements Be, Mg and Ca should display similar chemical and physical properties. They do. The elements Be, Mg, Ca fall under the group of alkaline–earth metals. All these elements are metallic in nature, their oxides are alkaline in nature and they have a valency of +2.
Now look at another vertical column that has carbon (C) as the first element. The eighth element from C is silicon (Si). It is seen that C and Si are similar in properties. Both of them show tetra–valence. They are non–metals and form oxides easily. Thus Newlands law of octaves hold good.
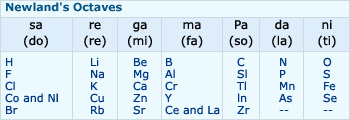 In order to fit elements into his table, Newlands adjusted two elements in the same slot, but also kept
some unlike elements under the same note. Cobalt and nickel are in same slot and are placed in the same column as fluorine,
chlorine and bromine which have very different properties. Iron, which resembles cobalt and nickel in properties, has been
placed far away from them. Thus, Newlands Law of Octaves worked well with lighter elements only.
In order to fit elements into his table, Newlands adjusted two elements in the same slot, but also kept
some unlike elements under the same note. Cobalt and nickel are in same slot and are placed in the same column as fluorine,
chlorine and bromine which have very different properties. Iron, which resembles cobalt and nickel in properties, has been
placed far away from them. Thus, Newlands Law of Octaves worked well with lighter elements only.
Newlands law of octaves failed for the following reasons:
- It was not valid for elements that had atomic masses higher than Ca.
- When more elements were discovered, such as elements from the noble gases such as He, Ne, Ar, they could not be accommodated in his table.
But the most important contribution in the process of classification of elements was the periodicity he saw in every eighth element. The modern periodic table drew heavily from the concept of periods of eight. Also it must be noted that Dobereiner's triads occurred in the octaves of Newlands.