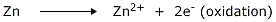Luigi galvani was an Italian physician and physicist who studied medicine. The electro chemical cell was named after him as galvanic cell for his contributions to electro chemistry and Simple galvanic cell.
Luigi galvani was an Italian physician and physicist who studied medicine. The electro chemical cell was named after him as galvanic cell for his contributions to electro chemistry and Simple galvanic cell.
The galvanic cell is a device used to convert the chemical energy produced in a redox reaction into electrical energy. The Galvanic cell, named after Luigi Galvani, consists of two different metals connected by a salt bridge or a porous disk between the individual half–cells. It is also known as a electrochemical cell.
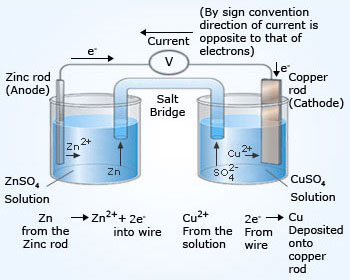
In a galvanic cell, chemicals present undergo oxidation and reduction (redox) reactions in order to generate electrons. This flow of electrons can be diverted to devices such as ipods, Laser pointer, cell phones, etc., a galvanic cell consists of two beakers connected with a salt–bridge. These beakers are known as cells. If the two cells with the reactants contain a metal that metal serves as a electrode for the cell. Usually an inert electrode made of platinum, silver or gold is used. These inert electrodes do not undergo reaction, but their surface merely acts as a surface at which oxidation and reduction occurs. These electrodes present in each cell, one with the same metal present in the reactants and one with the inert electrode are connected to each other with a metal wire, usually copper and a device such as meter (voltmeter or ammeter) to make the circuit complete.
 Working of Galvanic cell
Anode (negative terminal) is Zn (oxidation takes place).
Working of Galvanic cell
Anode (negative terminal) is Zn (oxidation takes place). 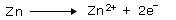
Cathode (positive terminal) is Cu (reduction takes place).
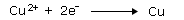
The electrode at which cations (metals with positive ions) get reduce is called as cathode. The electrode at which anions (metals with negative ions) get oxidized is called as anode. The device such as voltmeter consists of a positive side and a negative side. The positive side of voltmeter is connected to cathode and the negative side of voltmeter is connected to anode. The voltmeter shows readings related to the flow of electrons. The flow of charge is carried by electrons in the wires and by ions through the solutions and salt bridge. The chemicals present in the cell with an anode are written as an oxidation half–reaction. The chemicals present in the cell with a cathode are written as an reduction half–reaction.
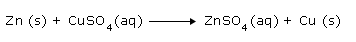
For Example: The redox reaction between Zn and CuSO4 is